Pharmaceutical Scientist / Technologist
Entry Level Qualification
Class 12
Career Fields
Information Technology & Computer Science
For Specially Abled








About Career
PARTICULARS | DESCRIPTION |
Name | Pharmaceutical Scientist / Technologist |
Purpose | Discover and innovate |
Career Field | Information Technology & Computer Science |
Required Entrance Exam | NEET UG, GPAT |
Average Salary | 300000 - 600000 Rs. Per Year |
Companies For You | Abbott Laboratories, Sanofi-Aventis, Pfizer & Many More |
Who is Eligible | Class 12th Pass |
Pharmaceutical Scientists/Technologists work to discover new drug molecules, discover new methods or systems for delivering drugs to the human body (such as tablets, liquids, injections, skin patches, gels, creams, etc.), and improve efficiency of drugs as well as that of medications or medicines.
Understand that drug molecules are responsible for therapeutic effects (therapeutic means cure or treatment of diseases or ailments), while medications or medicines are what we consume or delivered to our body. Typically, a medication or medicine is a formulation (such as a tablet or a liquid or an injection) which is a combination of drug molecules and other compounds (such as water, sugar powder, gelatine, and corn starch) which have no therapeutic effects.
Pharmaceutical Scientists/Technologists also:
1. Discover and innovate drugs and medicine manufacturing systems and processes;
2. Innovate, discover, and improve analytical methods for examining or assessment of the quality of drugs, medicine formulations, and other materials used in the manufacturing of drugs and medicines;
3. Research on Human Physiology, Pharmacology, Pharmacognosy, and Medicinal Chemistry as well as work in many research areas in life sciences including Microbiology, Biochemistry, and Biotechnology.
Now, before you read about what you will do as a Pharmaceutical Scientist/Technologist in several fields of work, you need to understand some terms and concepts
Active Pharmaceutical Ingredient (API): APIs contain the drug molecules which cure a disease or ailment. Ingredients other than the APIs in a medicine do not have a therapeutic (or curative) property.
Formulations: Formulations are what we consume as a medicine. We commonly refer to formulations as ‘medicines’ or ‘medications’. A formulation or a medicine contains several ingredients like API (Active Pharmaceutical Ingredient) or a combination of APIs along with several ingredients to give several properties to the medicine. For example, sugar could be added to give a sweet taste to a syrup. A binding agent could be added in a tablet. Distilled water or other liquids could be added to an injectable and so on.
Dosage forms or drug delivery systems: Drug delivery systems are ways of transporting a drug molecule into the human body. Overall, drug delivery systems consist of formulations, systems and technologies for making sure that a drug is transferred to the human body safely and in a way so that the molecule sustains the therapeutic (or curative) property of the drug.
Drug delivery systems may consist of dosage forms and systems like oral delivery (tablets, and liquids), intra-venous injectables and intra-muscular (injections), topical (gels, creams, skin patches, etc.), transmucosal like nasal and ocular (nasal drops, eye drops, etc.), inhalation (inhalers), etc.
Generic, Patented and OTC medicines: Generally, two kinds of drugs or medicines are available in the market. One is called ‘prescription drugs’. These drugs, as you can understand, are saleable only against a prescription by a registered medical practitioner or a doctor. The other kinds of drugs or medicines do not need a prescription to buy. These are called OTC or ‘Over The Counter’ drugs.
When a drug molecule is first developed, it is patented in the name of the company or the enterprise which has developed it. The patent gives the company or the enterprise the sole right to sell the drug across the world for a fixed number of years. Once the patent period is over, the drug is said to have become a generic drug and then it can be sold by any company, if approved by the drug control authorities in a country.
Biologic medicines and biosimilars: First of all, understand that the API in a formulation or a ‘medicine’ could be a synthetically produced chemical molecule or it may be derived from the living cells of an organism. If the API is the one which is derived from the living cells of an organism, then the medicine is called a biologic medicine. In characteristics, biological medicines consist of large molecules, much larger than a synthetic drug molecule. Biologic medicines include therapeutic proteins, DNA vaccines, etc.
In generic formulations (or what we commonly referred to as ‘generic medicines’) from different pharmaceutical manufacturers, the API (Active Pharmaceutical Ingredient as explained above) could be the same. But in case of a biological medicine, the active ingredient could be different from different manufacturers. Although the active ingredient could be ‘similar’ but they may differ in certain aspects. These are called ‘Biosimilars.’
So, what are be the various fields of work and what will you do in those?
Drug discovery research
Drug discovery involves – a) chemical research - molecular research, molecule synthesis, etc. to find out a new drug molecular entity, b) biological research – this includes biological assessment of the new molecular entities that are developed through chemical research. Or, simply put, assessment of the biological properties of the new drug molecule, and c) pharmacological and medicinal research – that is, understanding the disease curing properties of the newly developed drug molecule.
Clinical Pharmacology
Clinical pharmacology research establishes clinical usage of a newly discovered drug.
Clinical research
Clinical research determines the safety and efficacy (effectiveness) of the newly discovered drug molecule on a human body through pre-clinical trials and after the pre-clinical trials, through various stages or phases of clinical trials like I, II, III, and IV. In the pre-clinical trials, the newly discovered drug is tested on a non-human subject like a rodent and the clinical trials are done on humans, firstly on healthy volunteers and then on patients.
API research and development
Researching and developing newer Active Pharmaceutical Ingredient (API) which may be a newly developed drug molecule or a compound of the molecule or a combination of the new molecule along with other molecules or compounds.
Drug delivery research
Drug delivery includes research and development of new products, improving existing products, and expanding product application and drug delivery systems.
Formulation research
This involves research to find out the right formulation for the administration of a drug molecule.
Analytical research
Many chemical and biochemical processes are involved at all stages of drug discovery, clinical research, commercialization of the drug and manufacturing. Analytical research involves the development of chemical and biochemical methods and assessment for various processes during discovery, commercialization, manufacturing and quality control of drugs.
Bioinformatics/ Clinical Informatics / Pharmacoinformatics
Drug discovery processes and thereafter clinical research and clinical pharmacology produces humongous data. Bio- or clinical informatics analyses the data for various usages by the pharmaceutical manufacturers and regulatory bodies.
Pharmaceutical Process research and development research
This involves research for manufacturing processes in a commercial scale. Also involves research and development of chemical processes for generic drugs and intermediates.
Quality control and quality assurance
This involves quality checking and control to ensure a pre-determined level of quality in all the medical products manufactured.
You can also find work in biologics or biosimilars:
1. Research and development of biological molecules (also called biopharmaceuticals) and biosimilars.
2. Research and development of formulations containing the biopharmaceuticals and biosimilars.
3. Process research and development – involves research and development of chemical and biological/biotechnological processes for the manufacturing of biopharmaceuticals and biosimilars.
Besides the above, you may also find work opportunities in:
1. OTC Research and development – this involves product and process development and optimization, analytical development, technology adaptation, etc.
2. Regulatory affairs – this involves managing the regulatory processes in terms of meeting the norms prescribed by various global practices, drug administrators, etc.
Key Roles and Responsibilities:
In all the fields of research work as mentioned above, except in Manufacturing, Quality Control, Bioinformatics and Regulatory Affairs, you will have one or more of the following responsibilities:
1. Study the causes of various diseases and researching new drugs and drug delivery systems.
2. Improvement of the efficiency of existing formulations and delivery systems.
3. Documenting all the developments in experiments and researches.
4. Collaborate with different researchers, academics, scientists and research institutes to aid in the development of a drug.
5. Study journals and research materials of research being carried out in other companies and keep up to date with the developments.
6. Conducting experiments and analyzing results.
7. Writing papers and getting them published in pharmaceutical journals.
8. Provide analytical testing support at all stages of production.
In manufacturing function, you will have the following responsibilities:
1. You will be participating in or later supervising & managing the chain of activities/operations in a drug manufacturing plant engaged in manufacturing of pharmaceutical formulations, ayurvedic/herbal preparations, synthetic hormones, nutraceuticals or more.
2. You will be typically involved in following cGMP (current good manufacturing practices) in processing and production activities.
3. The environmental conditions, machine utilization records in the manufacturing area and other records pertaining to the various stages of manufacturing must be verified and monitored.
In quality control work areas, you will have the following responsibilities:
1. Testing of all laboratory samples including raw materials, in-process, finished products, validation, stability, and environmental.
2. Recording of analytical results accurately.
3. Operation, maintenance, and calibration of laboratory instruments.
4. Review and interpretation of data for conformance to procedures, standards and protocols and/or real-time recognition of aberrant data and results.
5. Assist in the improvement of quality systems by creating or revising worksheets and other lab documentation systems.
6. Comply with and implement safety standards.
In Bioinformatics, you will have the following responsibilities:
1. Create novel computational approaches and analytical tools as required by research goals.
2. Consult with researchers to analyze problems, recommend technology-based solutions, or determine computational strategies.
3. Analyze large molecular datasets such as genomic sequence data and proteomics data for clinical or basic research purposes.
4. Keep abreast of new biochemistries, instrumentation, or software by reading scientific literature and attending professional conferences.
5. Develop data models and databases.
6. Develop new software applications or customize existing applications to meet specific scientific project needs.
7. Design and apply bioinformatics algorithms including unsupervised and supervised machine learning, dynamic programming, or graphic algorithms.
In Regulatory Affairs, you will have the following responsibilities:
1. Coordinate, prepare or review regulatory submissions for domestic or international projects.
2. Review product promotional materials, labeling, batch records, specification sheets, or test methods for compliance with applicable regulations and policies.
3. Maintain current knowledge base of existing and emerging regulations, standards, or guidance documents.
4. Interpret regulatory rules or rule changes and ensure that they are communicated through corporate policies and procedures.
5. Determine the types of regulatory submissions or internal documentation that are required in situations such as proposed device changes or labeling changes.
6. Coordinate efforts associated with the preparation of regulatory documents or submissions.
Career Entry Pathway
Class 10 all subjects as per scheme of studies - Class 11-12 Physics, Chemistry along with Mathematics or Biology & any other subject as per scheme of studies –B.Pharm./B.Tech. in Pharmaceutical Sciences/Technology/Similar
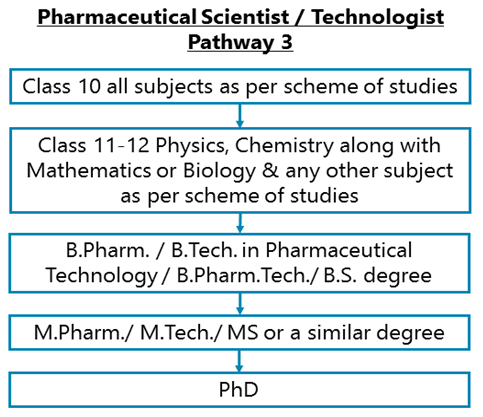
After completing Class 11-12 Physics, Chemistry along with Mathematics or Biology & any other subject as per scheme of studies, you can go for a Bachelor degree in Pharmacy, Pharmaceutical Science, Pharmaceutical Technology, or in a similar field. You may opt for a B.Pharm. / B.Tech. in Pharmaceutical Technology / B.Pharm.Tech./ B.S. degree.
Class 10 all subjects as per scheme of studies - Class 11-12 Physics, Chemistry along with Mathematics or Biology & any other subject as per scheme of studies –B.Pharm./B.Tech. in Pharmaceutical Sciences/Technology/Similar– M.Pharm. / M.Tech./ MS / similar degree or Integrated M.Pharm.-Ph.D. OR MS-Ph.D.
After completing Class 11-12 Physics, Chemistry along with Mathematics or Biology & any other subject as per scheme of studies, you can go for a Bachelor degree in Pharmacy, Pharmaceutical Science, Pharmaceutical Technology, or in a similar field. You may opt for a B.Pharm. / B.Tech. in Pharmaceutical Technology / B.Pharm.Tech./ B.S. degree. After your Bachelor’s degree, you may opt for an M.Pharm./ M.Tech./ MS or an integrated M.Pharm.-Ph.D. or an MS-Ph.D. in various specialized fields Pharmaceutical Science and Technology such as Pharmaceutics/ Pharmacology/ Pharmaceutical Sciences/ Medicinal Chemistry or Pharmaceutical Chemistry/ Pharmacognosy/ Pharmaceutical Biotechnology/ Pharmaceutical Engineering / Biochemistry/ Microbiology/ etc.
Class 10 all subjects as per scheme of studies - Class 11-12 Physics, Chemistry along with Mathematics or Biology & any other subject as per scheme of studies –B.Pharm./B.Tech. in Pharmaceutical Sciences/Technology/Similar– M.Pharm. / M.Tech./ MS / similar degree - Ph.D.
After completing Class 11-12 Physics, Chemistry along with Mathematics or Biology & any other subject as per scheme of studies, you can go for a Bachelor degree in Pharmacy, Pharmaceutical Science, Pharmaceutical Technology, or in a similar field. You may opt for a B.Pharm. / B.Tech. in Pharmaceutical Technology / B.Pharm.Tech./ B.S. degree. After your Bachelor’s degree, you may opt for an M.Pharm./ M.Tech./ MS or a similar degree in various specialised fields Pharmaceutical Science and Technology such as Pharmaceutics/ Pharmacology/ Pharmaceutical Sciences/ Medicinal Chemistry or Pharmaceutical Chemistry/ Pharmacognosy/ Pharmaceutical Biotechnology/ Pharmaceutical Engineering / Biochemistry/ Microbiology/ etc. After your M.Pharm./ M.Tech. / MS degree, you may do a Ph.D. in a specialised field of Pharmaceutical Sciences/ Technology such as the few which are mentioned above or in fields like Pharmacogenomics/ Clinical Pharmacology/ Pharmacovigilance and Regulatory Affairs/ Nutraceuticals/ Biopharmaceuticals/ Pharmaceutical Outcomes Research / Pharmacoinformatic/ etc.
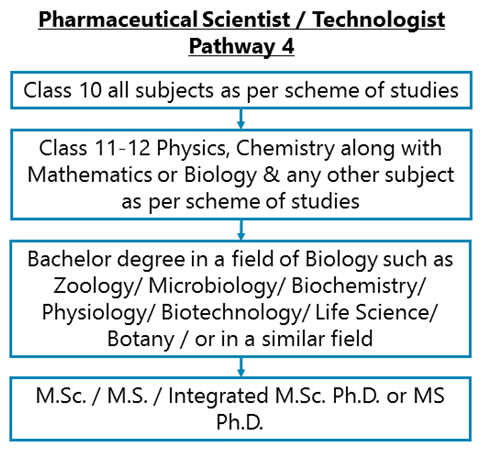
Class 10 all subjects as per scheme of studies - Class 11-12 Physics, Chemistry along with Mathematics or Biology & any other subject as per scheme of studies –B.Sc./ B.S. in Zoology/ Microbiology/ Biochemistry/ Physiology/ Biotechnology/ Life Science/ Botany / in a similar field – M.Sc. / M.S. / Integrated M.Sc. Ph.D. or MS Ph.D. in Zoology/ Microbiology/ Biochemistry/ Physiology/ Biotechnology/ Life Science/ Genetics/ Molecular Biology/ or in a similar field
After completing Class 11-12 Physics, Chemistry along with Mathematics or Biology & any other subject as per scheme of studies, you may opt for a Bachelor degree in a field of Biology such as Zoology/ Microbiology/ Biochemistry/ Physiology/ Biotechnology/ Life Science/ Botany / or in a similar field and then do a Master’s degree or an integrated Master’s-Ph.D. program in any of the fields mentioned above or in a field like Molecular Biology/ Biotechnology/ Genetics/ etc.
Class 10 all subjects as per scheme of studies - Class 11-12 Physics, Chemistry along with Mathematics or Biology & any other subject as per scheme of studies –B.Sc./ B.S. in Zoology/ Microbiology/ Biochemistry/ Physiology/ Biotechnology/ Life Science/ Botany / in a similar field – M.Sc. / M.S. in Zoology/ Microbiology/ Biochemistry/ Physiology/ Biotechnology/ Life Science/ Genetics/ Molecular Biology/ Botany or in a similar field - Ph.D. in various specialised areas such as Biopharmaceuticals, Pharmaceutical Chemistry, Pharmacognosy, Pharmacology, Pharmacogenomics, Pharmacoinformatic, Phytochemistry, Nutraceuticals, etc.
After completing Class 11-12 Physics, Chemistry along with Mathematics or Biology & any other subject as per scheme of studies, you may opt for a Bachelor degree in a field of Biology such as Zoology/ Microbiology/ Biochemistry/ Physiology/ Biotechnology/ Life Science/ Botany / or in a similar field and then do a Master’s degree in any of the fields mentioned above or in a field like Molecular Biology/ Biotechnology/ Genetics/ etc. Then you can do a Ph.D. in various specialised fields such as Biopharmaceuticals, Pharmaceutical Chemistry, Pharmacognosy, Pharmacology, Pharmacogenomics, Pharmacoinformatic, Phytochemistry, Nutraceuticals, etc.
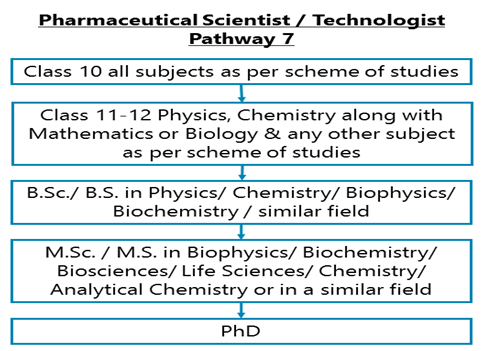
Class 10 all subjects as per scheme of studies - Class 11-12 Physics, Chemistry along with Mathematics or Biology & any other subject as per scheme of studies –B.Sc./ B.S. in Physics/ Chemistry/ Biophysics/ Biochemistry / similar field – M.Sc. / M.S. / Integrated M.Sc. Ph.D. or MS Ph.D. in Biophysics/ Biochemistry/ Biosciences/ Life Sciences/ Chemistry/ Analytical Chemistry or in a similar field
After completing Class 11-12 Physics, Chemistry along with Mathematics or Biology & any other subject as per scheme of studies, you may opt for a Bachelor degree in Physics/ Chemistry/ Biophysics/ Biochemistry/ or in a similar field. Then you can do a Master’s degree or an integrated Master’s-Ph.D. degree in Biophysics/ Biochemistry/ Bioscience/ Life Science/ Analytical Chemistry/ or in a similar field.
Class 10 all subjects as per scheme of studies - Class 11-12 Physics, Chemistry along with Mathematics or Biology & any other subject as per scheme of studies –B.Sc./ B.S. in Physics/ Chemistry/ Biophysics/ Biochemistry / similar field – M.Sc. / M.S. in Biophysics/ Biochemistry/ Biosciences/ Life Sciences/ Chemistry/ Analytical Chemistry or in a similar field – Ph.D.
After completing Class 11-12 Physics, Chemistry along with Mathematics or Biology & any other subject as per scheme of studies, you may opt for a Bachelor degree in Physics/ Chemistry/ Biophysics/ Biochemistry/ or in a similar field. Then you can do a Master’s degree in Biophysics/ Biochemistry/ Bioscience/ Life Science/ Analytical Chemistry/ or in a similar field. After your Master’s degree, you can do a Ph.D. in any of the above mentioned fields.
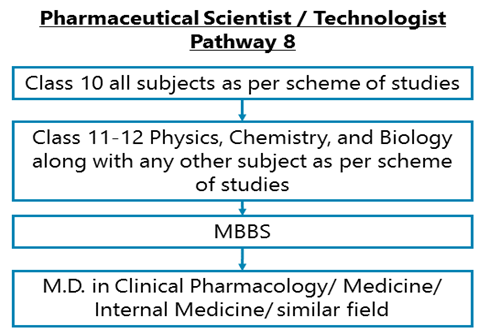
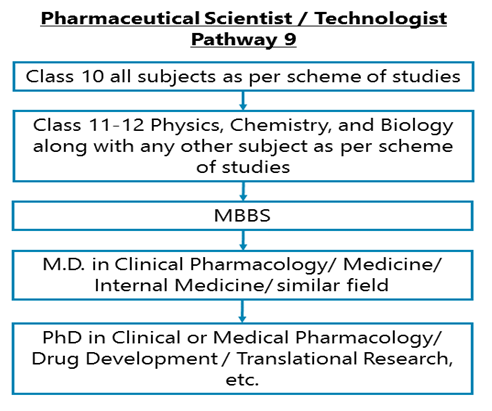
Class 10 all subjects as per scheme of studies - Class 11-12 Physics, Chemistry along with Biology & any other subject as per scheme of studies –MBBS – M.D. in Clinical Pharmacology/ Medicine/ Internal Medicine/ similar field
After completing Class 11-12 Physics, Chemistry, and Biology along with any other subject as per scheme of studies, you may opt for a Bachelor degree in Medicine and Surgery followed by an M.D. in Clinical Pharmacology/ Medical Pharmacology/ Medicine / Internal Medicine / or in a related field.
Class 10 all subjects as per scheme of studies - Class 11-12 Physics, Chemistry along with Biology & any other subject as per scheme of studies–MBBS – M.D. in Clinical Pharmacology/ Medicine/ Internal Medicine/ similar field – Ph.D.
After completing Class 11-12 Physics, Chemistry along with Biology & any other subject as per scheme of studies, you may opt for a Bachelor degree in Medicine and Surgery followed by an M.D. in Clinical Pharmacology/ Medical Pharmacology/ Medicine / Internal Medicine / or in a related field. After your Master’s degree, you can do a Ph.D. in Clinical or Medical Pharmacology/ Drug Development / Translational Research, etc.
Class 10 all subjects as per scheme of studies - Class 11-12 Physics, Chemistry, and Mathematics along with any other subject as per scheme of studies–B.Sc./B.S. in Statistics/ Mathematics/ B.Stat./ B.Math./ similar degree – M.Sc./ M.S. in Biostatistics / Bioinformatics/ similar field – Ph.D. in Biostatistics/ Bioinformatics/ Pharmacoinformatic/ similar field
After completing Class 11-12 Physics, Chemistry, and Mathematics along with any other subject as per scheme of studies, you may opt for a Bachelor degree in Statistics/ Mathematics followed by a Master’s degree in Biostatistics/ Bioinformatics/ in a similar field. After your Master’s degree, you may do a Ph.D. in Biostatistics/ Bioinformatics/ Pharmacoinformatic/ in a similar field.
Class 10 all subjects as per scheme of studies - Class 11-12 Physics, Chemistry, and Mathematics along with any other subject as per scheme of studies –B.E./ B.Tech./ similar degree in Biochemical Engineering/ Biotechnology/ Chemical Engineering/ similar field – M.E./ M.Tech. /M.S. in Biochemical Engineering / Biotechnology/ Pharmaceutical Engineering/ Pharmaceutical Biotechnology/ similar field
After completing Class 11-12 Physics, Chemistry, and Mathematics along with any other subject as per scheme of studies, you may opt for a Bachelor degree in Biochemical Engineering / Biochemical Engineering & Biotechnology/ Biotechnology/ Chemical Engineering/ similar field. You can then do an M.E./ M.Tech/ MS or a similar degree in Biochemical Engineering / Biotechnology/ Pharmaceutical Engineering/ Pharmaceutical Biotechnology or a similar field.
Class 10 all subjects as per scheme of studies - Class 11-12 Physics, Chemistry along with Mathematics or Biology & any other subject as per scheme of studies – Pharm.D.
After completing Class 11-12 Physics, Chemistry along with Mathematics or Biology & any other subject as per scheme of studies, you can go for a 6 year Pharm.D. course. This involves 5 years of study and 1 year of internship or residency at a hospital. This pathway prepares you for a role in Clinical Pharmacy/ Hospital Pharmacy practice.
Class 10 all subjects as per scheme of studies - Class 11-12 Physics, Chemistry along with Mathematics or Biology & any other subject as per scheme of studies – B.Pharm./ similar Bachelor’s degree in Pharmaceutical Sciences –Phar.D. Post Baccalaureate course
After completing Class 11-12 Physics, Chemistry along with Mathematics or Biology & any other subject as per scheme of studies, you can go for a Bachelor degree in Pharmacy (B Pharm.) or a similar degree course and then do a 3 year Phar.D. Post Baccalaureate course. This involves 2 years of study and 1 year internship or residency at a hospital.This pathway prepares you for a role in Clinical Pharmacy/ Hospital Pharmacy practice.
Required Qualification & Competencies
To get into this field, you will need to pass the Class 10 examination all subjects as per scheme of studies. Then you must complete Class 11-12 Physics, Chemistry, along with Mathematics, Biology & any other subject as per scheme of studies.
Then you can do a degree in Pharmacy or in any one of the following fields. Master’s degree or higher is preferred if you are interested in R&D or manufacturing roles. You can study for a Bachelor’s / Master’s / Doctoral degree in any of the following fields (Note that all these fields may not offer you a degree at all three levels, that is in Bachelor’s, Master’s and Doctoral. Some fields may offer a degree only at the Master’s or at the Doctoral level):
1. Pharmaceutical Sciences
2. Chemical and Pharmaceuticals Technologies
3. Pharmaceutical Biotechnology (Engineering)
4. Pharmaceutical Technology
5. Pharmaceutical Biotechnology
6. Pharmaceutical Regulatory Affairs
7. Pharmaceutics
8. Pharmacy Administration, Economics & Outcomes Sciences
9. Pharmacy Practice
10. Pharmaceuticals & Fine Chemicals
11. Pharmaceutics & Pharmaceutical Engineering
12. Bhaishajya Kalpana (Pharmaceutics)
13. Dravya Guna (Pharmacology)
14. Clinical Pharmacology
15. Medical Pharmacology
16. Biopharmaceuticals Sciences & Technology
17. Pharmaceutical and Medicinal Chemistry
18. Pharmacogenomics & Genetics
19. Pharmacognosy
20. Pharmacoinformatic
21. Pharmacology
22. Quality Assurance (Pharmaceuticals)
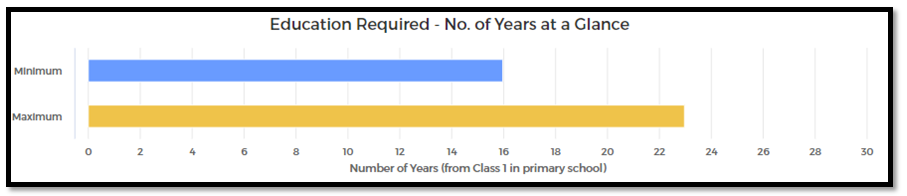
MINIMUM EDUCATION REQUIRED | MAXIMUM EDUCATION REQUIRED |
Under Graduate | Post-Doctoral |
COMPETENCIES REQUIRED
Interests
1. You should have interests for Investigative Occupations. Investigative occupations involve working with ideas and quite a lot of thinking, often abstract or conceptual thinking. These involve learning about facts and figures; involve the use of data analysis, assessment of situations, decision making and problem-solving.
2. You should have interests for Conventional Occupations. Conventional occupations involve repetitive and routine tasks as well as fixed processes or procedures for getting things done. These occupations involve working more with data, systems, and procedures and less with ideas or creativity.
3. You should have interests for Social Occupations. Social occupations involve helping or assisting others; these involve working with and communicating with people to provide various services; these may involve educating and advising others.
Knowledge
1. You should have knowledge of Biological Sciences - plants and animals, their anatomical structure, cell structure, tissues, physiological functions, evolution, and all other related aspects.
2. You should have knowledge of Production and Processing - raw materials, production machinery, production systems, production processes, quality control, and other techniques for manufacturing and distribution of drugs.
3. You should have knowledge of Pharmacology and Pharmaceutical Sciences – about various drugs, chemical properties of the drugs, therapeutic properties of the drugs, how they work in human or animal body, how drugs are manufactured, etc.
Skills
1. You should have Active Learning Skills - Focused and continuous learning from various sources of information, observation and otherwise for application in getting work done.
2. You should have Scientific Skills - using various scientific rules and methods to get things done or solve problems.
3. You should have Technical Skills - using various technologies and technical methods to get things done or solve problems.
4. You should have Critical thinking Skills –analysis of complex situations, using logic and reasoning to understand the situations and take appropriate actions or make interpretations and inferences.
5. You should have Coordination Skills - working together with other people to get things done.
6. You should have Operating Skills - operating various machines, equipment, devices and systems.
7. You should have Judgment and Decision Making Skills - considering pros and cons of various decision alternatives; considering costs and benefits; taking appropriate and suitable decisions.
8. You should have Quality Control Analysis Skills - conducting tests and inspections of products, services, or processes to evaluate quality or performance.
Ability
1. You should have Problem Solving Ability - analysis and understanding of problems, evaluating various options to solve the problems and using the best option to solve the problems.
2. You should have Inductive Reasoning Ability - The ability to combine pieces of information from various sources, concepts, and theories to form general rules or conclusions. For example, analysing various events or situations to come out with a set of rules or conclusions.
3. You should have Originality - The ability to come up with unusual or innovative ideas about a given topic or situation, or to develop creative ways to solve a problem.
4. You should have Verbal Reasoning Ability - The ability to think and reason with words; the ability to reason out ideas expressed in words.
5. You should have Written Expression Ability - The ability to communicate information and ideas in writing so others will understand.
6. You should have Deductive Reasoning Ability - apply general rules and common logic to specific problems to produce answers that are logical and make sense. For example, understanding the reasons behind an event or a situation using general rules and common logic.
7. You should have Speed of Closure - The ability to quickly make sense of, combine, and organize information into meaningful patterns.
Personality
1. You are always or mostly careful about your actions and behavior.
2. You are always or mostly organized in your day-to-day life and activities.
3. You are always practical or in most situations.
4. You always feel secure in your surroundings and in most situations.
5. You are always calm or generally remain calm in most situations.
6. You can always act independently or could do so in most situations.
Career - Job Opportunities & Profiles
Job opportunities could be across various functions of the pharmaceutical industry and research organizations. Following are the various opportunities:
1. After your Bachelor’s / Master’s degree, you may get an opportunity as a Manufacturing/ Production Executive/ Assistant Manager-Production/ in a similar role or you may get a role as a Quality Control Chemist/ Quality Assurance Chemist/ or a similar role with a pharmaceutical company in their manufacturing department. After your Master’s degree, you may also get offers in a role in Pharmaceutical Regulatory Affairs function in a pharmaceutical company.
2. After your Bachelor’s degree, you may find a job with the Central Drugs Standard Control Organisation (CDSCO) or a State Drug Control Organisation as an Assistant Drug Inspector / Research Assistant.
3. After your Master’s degree, you may get an opportunity as a Research Executive/ Research Associate/ in a similar role at the R&D (Research & Development) functions of various pharmaceutical companies (which are involved in drug development, manufacturing, and sale of medicines).
4. After your Master’s degree, you may get an opportunity in a pharmaceutical research organisation as a Research Associate (this is a project based appointment)/ Junior Scientist (this is a permanent appointment).
5. After your Master’s degree, you may get an opportunity in a pharmaceutical research organisation or in a University as a Doctoral Fellow / Junior Research Fellow if they offer a Ph.D. program.
6. After your Master’s degree, you may an opportunity with a college/ University which offers courses in Pharmacy/Pharmaceutical Sciences/Technology. But for University positions, having a Ph.D. will be made mandatory soon.
7. After your Ph.D., you may get an opportunity in a pharmaceutical research organisation or in a University as a Post-Doctoral Fellow.
8. After your Ph.D., you may get an opportunity as an Assistant Professor in a University which offer courses in Pharmacy/ Pharmaceutical Sciences.
9. After your Ph.D. / or after your Ph.D. and experience as a Post-Doctoral Fellow, you may get a good opportunity in research and development with a pharmaceutical company or with a pharmaceutical research organisation or with a university. In these opportunities, you will begin as a Scientist/ Assistant Professor.
Few of the pharmaceutical companies (which produce formulations or medicines that we take) where you may find opportunities:
Abbott Laboratories, Sanofi-Aventis, Pfizer, Glaxo SmithKline, Zydus Cadilla, Wockhardt, Torrent Pharmaceuticals, Cipla, Dr. Reddy’s Laboratory, Lupin, SunPharmaceuticals, Cadilla Healthcare, Piramal Healthcare, Dabur, Glenmark Pharmaceuticals, Biocon, Aurobindo Pharma, IPCA, JubilantLifescience, Merck, Novartis, etc.






Few pharmaceutical companies which produce and market bulk drugs / API:
Bayer Pharmaceuticals, Sandoz, Mylan, Hikal, Shasun, Adani Pharma, Unilab, Surya Pharmaceuticals, Dishman Pharmaceuticals, Hindustan Antibiotics, Bengal Chemicals and Pharmaceuticals, Apex Drugs, Eskay Fine Chemicals, Dhanuka Laboratories, Unimark Remedies, Rajasthan Drugs and Pharmaceuticals Ltd., Indian Drug and Pharmaceuticals Ltd., etc.
Few pharmaceutical companies which produce OTC (Over-The-Counter drugs):
Himalaya Drugs, P & G, Johnson & Johnson, Rekitt Benckiser, Dabur, Zydus Cadilla, Dishman
Pharma; Unichem Lab; Emami, TTK Healthcare, Zandu Pharma; many of the other
formulation companies listed above also produce OTC drugs.




Few research institutions where you may find jobs:
1. National Institute of Pharmaceutical Education and Research (NIPER)
2. Central Drugs Research Institute (CDRI), Lucknow
3. Central Institute of Medicinal and Aromatic Plants (CIMAP), Lucknow
4. Indian Institute of Toxicological Research (IITR), Lucknow,
5. Indian Institute of Integrative Medicine (IIIM), Jammu,
6. Institute of Integrative and Genomic Biology (IIGB), Delhi
7. Indian Institute of Chemical Technology (IICT), Hyderabad
8. Centre for Cellular and Molecular Biology (CCMB), Hyderabad
9. Indian Institute of Chemical Biology (IICB), Kolkata
10. North East Institute of Science and Technology(NEIST), Jorhat
11. National Institute of Immunology (NII), New Delhi
12. National Institute of PlantGenome Research (NIPGR), New Delhi
13. National Institute of Malaria Research (NIMR), Delhi, National Institute of Pathology (NIOP), Delhi
14. National Institute of Cholera and Enteric Diseases (NICED), Kolkata
SPECIALISATION TRACKS IN THIS CAREER
Pharmaceutical Scientist (Pharmacology)
Pharmacologists study actions of drugs on living systems. As for examples, they focus on how a drug affects physical, emotional & psychological wellbeing of the recipient, the type of drug being used, modes of administration, how the drug is absorbed as well as the characteristics of the person taking the drug.
Pharmaceutical Scientist (Pharmacognosy)
Pharmacognosy is the study and analysis of the biological, chemical, biochemical, and physical properties of medicines or crude drugs produced from natural sources such as plants, microbes, and animals. Many important drugs, including morphine, atropine etc., have originated from natural sources and continue to be good model molecules in drug discovery. Scientists practicing in this branch also deal with the history, cultivation, collection, extraction, isolation, bio assaying, quality control, and preparation of crude drugs of natural origin.
Pharmaceutical Scientist (Biopharmaceuticals)
Biopharmaceuticals include therapeutic proteins, fusion proteins, monoclonal antibodies, and antibody-drug conjugates which are all large complex molecules, > 1000 times bigger than 'conventional' small molecule drugs. They are more commonly called ‘biologics’. They are composed of sugars, proteins, or nucleic acids or complex combinations of these substances, or may be living cells or tissues manufactured in, extracted from, or semi synthesized from biological sources or isolated from living sources such as human, animal, plant, fungal, or microbial. Pharmaceutical Scientists practicing in this domain are usually PhDs. They discover such drugs by identifying bio-molecular processes inside the human body and simultaneously mapping it to the source cell.
Pharmaceutical Scientist (Pharmaceutical and Medicinal Chemistry)
These mainly combine the two arms of Science namely Pharmacology and Organic Chemistry (mainly synthetic organic chemistry). Pharmaceutical Scientists in this field are involved with drug design, chemical synthesis and development for pharmaceutical agents or bio-active molecules.
Pharmaceutical Scientist (Pharmacoinformatic)
Pharmacoinformatic Scientists focus on effective use of information technology (databases, digital tools / software and online repositories) for drug discovery. The broad sub-classifications within this branch of study are many, including immunoinformatic (using databases and software for modeling & informatics of the immune system and study of immunological macromolecules to address important questions in immunobiology / vaccinology), genomatics (analyzing / extracting / computing biologically relevant information from the rapidly growing biological and essential gene sequence databases), neuromatics (digital organization and analysis of medical data pertaining to neurosciences), toxicoinformatics (for the prediction of toxicity of chemical molecules planned to be used in drugs), metabolomatics (study of the disposition of a pharmaceutical compound within an organism with the help of digital tools), healthinformatics etc. All of these are tied to decision making in the pharmaceutical industry and feedback received in response to a drug so that future designing of the drugs can be made to suit the needs of the patients.
Pharmaceutical Scientist (Pharmaceutical Regulatory Affairs)
For getting a newly discovered drug into the market and even for selling any other drug in the market of a country, approval from the drug regulatory authority of the country is required. Without approval of such an agency, no drug could be sold. For example, in India, there is the Central Drugs Standard Control Organization (CDSCO) and in the USA, there is the FDA (Food and Drug Administration). Pharmaceutical Scientists working in regulatory affairs prepares applications for approval of various drugs from the drug control authorities; manage the entire process of approval of drugs on behalf of a company; manage all affairs related to compliance to a country's laws, rules, and regulation related to manufacturing, sales, and dispensation of drugs in a country.
Pharmaceuticals Technologist (Pharmaceutics/Pharmaceutical Engineering)
Tablets for oral consumption of medication have been utilized since approximately 1500 B.C., which were designed by predecessors of Pharmaceuticals Technologists! Now, Pharmaceuticals Engineers are using techniques focused on discovering, formulating, and manufacturing medication to make them more effective. For an example, think of Salvarsan (intravenous injection to treat a common bacterial disease) was chemically engineered from Atoxyl that is an arsenic containing compound which is actually harmful for humans but the structure of it was modified to make it kill the infection instead of affecting normal healthy human cells. Then another example, would be the invention of controlled release (sustained release over a 12-hour to 24-hour period) instead of immediate-release (all the medication is released in the body at once by the drug). Pills were designed in such a way that they could release the active ingredient through mechanisms such as osmosis and diffusion over a longer period while traveling through our bodies. The controlled release timescale can be extended to the order of months too!
Pharmaceutical Scientist (Pharmaceutical Quality Assurance)
Quality Assurance officers manage activities and responsibilities required to ensure that the drug that reaches a patient is safe, effective, and acceptable to the patient. Quality control is a critical function of a public pharmaceuticals supply business and vital to the successful development, manufacturing, and use of drugs. It emphasizes both the technicalandmanagerial actions to ensure medicine quality. Every manufacturing firm mandatorily has a QC window that deals with medicine sampling, specifications, testing, and the organization’s release procedures. This ensures that materials are released for sale / supply / use only after their quality has been judged as satisfactory. The Drug and Therapeutics Committees (DTC) of individual health care organizations are responsible for such evaluation of new medicines before they are added to the formulary.
Pharmaceutical Technologist (Pharmaceuticals & Fine Chemicals)
Most drugs are conjugates meaning they are a combination of an active ingredient and inactive ones. The inactive ones do not increase or affect the therapeutic action of the active ingredient and are typically dyes, binding materials, preservatives, and flavoring agents. Both the active and inactive drug components are basically chemical compounds. Manufacturers of fine chemicals or drug APIs (active pharmaceutical ingredients) are the major players in the innovative drugs and fine chemicals market today. Professionals working with such companies are majorly involved in production divisions or quality assurance wings or even R&D of drug design.
Pharmaceutical Scientist (Pharmacogenomics & Genetics)
Scientists in this field work towards the development of effective, safe medications and doses that will be tailored to a person's genetic makeup. Genes affect a person's response to drugs and this necessitates the study of pharmacology & genetics (pharmacogenomics to be more apt) as the current industry standard of “one size fits all” doesn’t work the same way for everyone. It can be difficult to predict who will benefit from a medication, who will not respond at all, and who will experience negative side effects. Researchers practicing in this domain try to learn how inherited differences in genes affect the body’s response to medications. This field however is still in its infancy. Its use is currently quite limited, but new approaches are under study in clinical trials.
Pharmaceutical Technologist (Pharmaceutical Biotechnology)
This branch is closely tied to the biopharmaceuticals domain in which large biotechnology corporations operate to innovate and manufacture drugs based on human, animal, plant, fungal, or microbial sources. It is a relatively new and emerging field in which the principles of biotechnology are applied to gene therapy, the development of drugs as well as genetic testing. Professionals in this field should be competent with hands-on training in recombinant DNA technology, basic molecular biology techniques, animal tissue culture, cell and molecular biology, drug discovery, fermentation, proteomics, diagnostic tests (ELISA), Western blot, nano-biotechnology etc. For an example, Eli Lilly’s Humulin, the first human insulin, targeted at diabetics, engineered through recombinant DNA technology, was invented in 1982 and approved by FDA (USA).
Pharmaceutical Scientist (Phytochemistry)
The prefix Phyto refers to ‘plants’. Scientists practicing in this domain focus on the area of discovery and development of herbal drugs / phytopharmaceuticals of terrestrial, microbial and marine origin, their quality control and analysis; understanding the synergy between components of a medicinal plant at a molecular level; production of drugs using plant tissue culture and fermentation technologies; studies on herbal cosmetics, medicinal plants etc. as well as screening of new drug molecules, standardized herbal drugs/extracts for their bioactivities etc.
Pharmaceutical Scientist (Nutraceuticals)
Nutraceuticals Scientists are involved in studying “a food item or parts of food items that provide medical or health benefits, including the prevention and treatment of disease”. These may range from herbal products, isolated nutrients, dietary supplements to processed products such as cereals and beverages. These in general contain lipids, vitamins, carbohydrates, proteins, minerals, or other necessary nutrients and are marketed in concentrated forms as capsules, pills, powders / tinctures either as a single substance or as combination preparations.
Pharmaceutical Scientist (Clinical Pharmacy/Pharmacy Practice)
This field mainly includes professionals working at hospitals / nursing homes / healthcare centers and similar patient care centers. Professionals in this field are experts in the therapeutic use of medicines or medications. They advise patients as well as other healthcare professionals about the best use of various medicines and drug delivery methods for earliest recovery of a patient from a disease or ailment. They advise about the various side effects of medicines and suggest alternative medicines to do away with the side effects. They may also suggest alternative medicines to lower the cost of treatment. They are often responsible for clarification of the drug-related queries of patients and play a very critical role in counseling patients regarding drug therapies. They need to interpret physicians’ orders and thereby detect therapeutic incompatibilities or suggest similar drugs.
Pharmaceutical Scientist (Pharmacy Administration, Economics & Outcomes Sciences)
This is a vast field composed of different functional segments that deal with distinct organizational processes to add value to a drug supply business. Especially focused on the business expansion area, this field involves professionals who study / analyze / compute the various possibilities or metrics of a new drug or new molecular entity (NME) to succeed in a regional market. This also deals with learning about the competition that a drug may or not face in the new market. They project such learnings before or during the launch of a new product or to assist in the R&D studies while a new drug is being designed. It also involves deriving knowledge through various international medical conferences or science congresses where researchers present their findings and drug manufacturers follow these to produce their next innovation. This branch is a relatively new industry segment and is of exceptional importance owing to the advent of too many players in the healthcare industry.
CAREER GROWTH
From entry level positions such as Assistant / Executive / Officer / Associate / Scientist / Technologist / Analyst etc., you may grow through various roles like Senior Scientist / Principal Scientist; Manager / Outpatient Pharmacy Manager; Senior Manager; Plant Head / Production Manager; General Manager; Division Head / Cluster Head / R&D Head; Global CI Manager; Head-BD & Marketing; Vice President (Plant Operations) / VP-Formulation Development; Chief Scientific Officer / Chief Operating Officer / Chief Health Officer and so on.
Remember, your growth, as well as your designations, will only depend on the functional division that you are associated with, so as mentioned here, if you are working in the R&D department you can be promoted to the position of Senior Scientist, Principal Scientist etc. or if you are in the Production & Processing division you can be promoted to Plant Head, Production Manager eventually in say not less than a decade and you can rise upwards from there.
Salary Offered
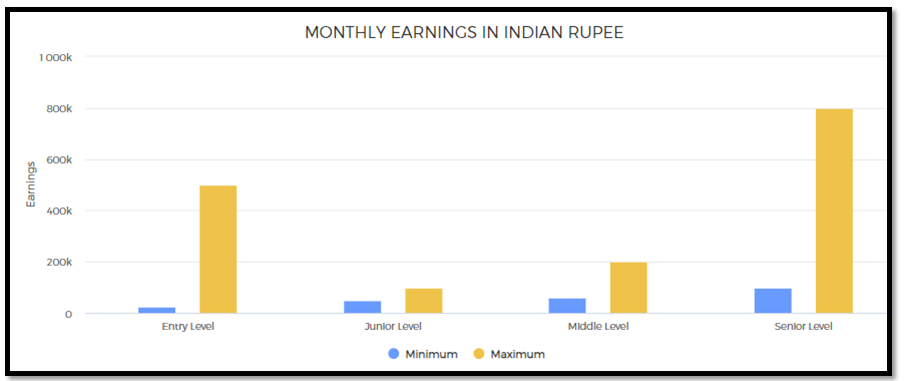
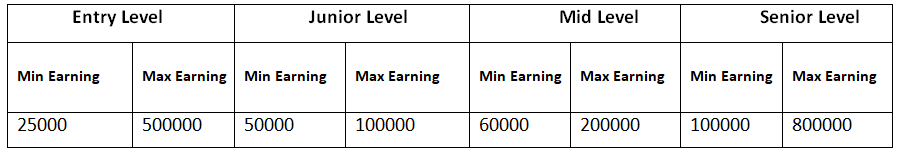
1. After your Bachelor’s degree, at entry-level jobs as a Trainee or Intern, you may expect to make about Rs. 25,000 – 50,000 or even more a month. Higher salaries are paid to graduates from premier institutions.
2. At the junior level jobs, you may expect to get about Rs. 50,000 – 1,00,000 or even more per month.
3. In mid-level jobs in India, (with 5-12 years of experience) may vary from Rs. 60,000 – 2,00,000 per month or even more.
4. At senior level (with 12-20 years of experience), you may earn about Rs. 1,00,000 – 8,00,000 or even more a month. Chief Scientific Officers may earn about Rs. 50,00,000 to 3,00,00,000 or much more per year.
Global (US)
1. At Entry level as an Intern or Trainee, you may earn about $4,000 - $7,000 per month.
2. At Junior-level, with an experience of 1-5 years, you may earn around $5,000 - $9,500 or more per month.
3. At Mid-level, with an experience of 6-12 years, you may earn around $6,500 - $10,500 per month.
4. At Senior-level, with an experience of 13-20 + years, you may earn around $ 8,500 - $12, 000 per month.
MONTHLY EARNINGS IN INDIAN RUPEE
1. Entry level: 0 - 2 years of work experience
2. Junior Level: From 1 to 12 years of work experience
3. Mid Level: From 5 to 20+ years of work experience
4. Senior Level: From 10 to 25+ years of work experience (there could be exceptions in some high-end technical, financial, engineering, creative, management, sports, and other careers; also in the near future, people will reach these levels much faster in many careers and in some careers, these levels will have no meaning as those careers will be completely tech skill driven such as even now, there is almost no level in a Cyber Security Expert’s job)
Work Activities
1. Analysing and interpreting data and information - Analysis of data and information to find facts, trends, reasons behind situations, etc.; interpretation of data to aid in decision making.
2. Assessing and determining compliance with standards, laws, rules and guidelines - Using relevant information, auditing information, processes and systems to determine whether organisations or people are complying with standards, laws, rules and guidelines.
3. Assessing and evaluating quality - Assessing and evaluating quality of materials, systems, processes, products, services, etc.
4. Calculating and computing - Calculating or computing using various mathematical formula and functions using computers or otherwise.
5. Communicating with co-workers and others - Communicating with people in writing, verbally or otherwise inside your workplace and various other people who have professional relationships with your place of work including vendors, government officials, etc. or with people at large.
6. Computing - Computing using various computer software applications; using software applications for scientific and technical work.
7. Creative thinking - Developing new ideas, concepts, innovative solutions to problems, newer ways of getting things done, designing products and services, etc.
8. Estimating quantity, cost, time and resources - Estimating sizes, volumes, distance, and quantity; estimating and determining time, costs, and resources; estimating materials required to perform a task.
9. Getting Information and learning - Observing, hearing, reading, using computers, or otherwise obtaining information and learning from it.
10. Making decisions and solving problems - Analysis of data and information; evaluation of alternative decisions and results of decisions; taking the right decisions and solving problems.
11. Managing and supervising - Managing and supervising work of others; setting goals; giving instructions; monitoring work performance, etc.
12. Operating machines, equipment, devices, systems and processes - Using various methods and techniques to operate machines, equipment, devices, systems and processes in industrial and other settings.
13. Organising, planning and prioritizing tasks - Planning and organising tasks in order to achieve work goals; prioritizing tasks to achieve goals and making the best use of the time available.
14. Processing information - Searching, compiling, tabulating, calculating, auditing, verifying or otherwise dealing with information processing including data entry, transcription, recording, storing and maintaining databases.
15. Providing advices and consultation to others - Giving advices or consultation to others about various issues, conceptual matters, know-hows, scientific matters, products or services.
16. Scheduling tasks - Scheduling project timelines, tasks and activities.
17. Strategic planning - Developing visions and goals, developing strategies and action plans for achieving visions and goals.
18. Updating and using relevant knowledge - Keeping updated with the latest knowledge relevant to your fields of work and use of the relevant knowledge in getting things done.
19. Using computers for work - Using computers for day-to-day office work; using computer software for various applications in day-to-day professional work; entering data and process information; for writing.
20. Working in a team - Working in a team of people; developing team; maintaining professional relationships among team members.
Future Prospects
You can expect a decent future in this field as the industry growth prospects are encouraging. The pharmaceutical industry is oriented toward extremely high-quality research that leads to new treatments for people in need. Indian pharmaceutical industry is a prominent industry in terms of production, consumption and exports of pharmaceutical products. By 2025, medicine spending in India is expected to grow by almost 9 to 12% making India one of the world’s top ten in this zone. India’s pharmaceutical industry is projected to expand at a rate of 22.4% compounded annually. This should take projected turnover to the mark of 55 billion US Dollars in terms of pharmaceutical production.
Exports of pharmaceutical products from India are projected to grow at 7.98% compounded annually, in value terms. Indian government is also preparing for a huge multi-billion dollar investment with 50 per cent public funding through its public private partnership (PPP) model, in order to enhance India’s innovation capability. The aim is to push India into top 5 pharmaceutical innovation hubs by 2020 and to establish global presence by launching 1 out of every 5-10 drugs discovered in India at global level by 2020.
India should be the 6th largest market globally in terms of absolute size. Presently, the industry is expanding at a healthy rate of 9-10% year-on-year. Various multinational companies like Novartis, Merck Sharp & Dohme are launching patent-protected drugs in India at price points lower by almost 20% than those in developed markets.
FUTURE PROSPECTS AT A GLANCE